 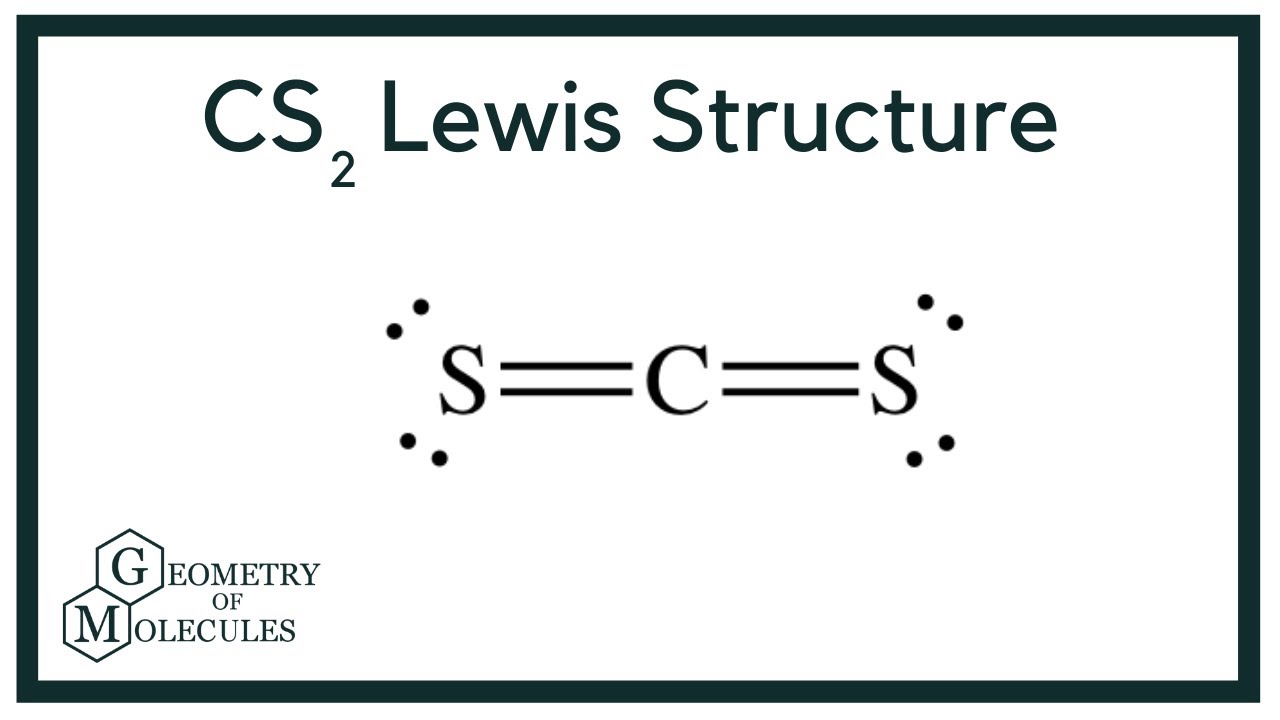
Ĭhen X, Li J, Jia C (2019) Thermodynamic properties of gaseous carbon disulfide. Just toxic or also bioregulatory and/or therapeutic? Chem Soc Rev 46:21–39. ĭeMartino AW, Zigler DF, Fukuto JM, Ford PC (2017) Carbon disulfide. Zhang X, Ponce V, Galvez-Aranda DE, Zhou G, Zhou H, Seminario JM (2021) CS 2 removal from C 5 distillates by reactive molecular dynamics simulations. Yue X, Wang H, Kong J, Li B, Yang J, Li Q, Zhang J (2020) A novel and green sulfur fertilizer from CS 2 to promote reproductive growth of plants. Liu N, Song X, Wang C, Li K, Ning P, Sun X, Wang F, Ma Y (2020) Surface characterization study of corn-straw biochar catalysts for the simultaneous removal of HCN, COS, and CS 2. Zeng Z, Dlugogorski BZ, Oluwoye I, Altarawneh M (2019) Combustion chemistry of carbon disulphide (CS 2). Wang W, Fan L, Wang G (2017) Carbon disulfide (CS 2) adsorption and dissociation on the Cu(100) surface: a quantum chemical study. Our results might contrite to provide a novel method to study the catalytic hydrolysis of CS 2. During the hydrolysis of CS 2, the five reaction paths are parallel and competitive. It was sought out that the hydrolysis of CS 2 was an exothermic reaction, and the increment in temperature was adverse to the reaction. Besides, to further clarify which reaction path is better, the hydrolysis kinetics of CS 2 was analyzed. Apart from this, to further explore the reaction of CS 2 hydrolysis, the natural bond orbital (NBO) analysis of the transition states was carried out. Similarly, the order of the barriers for the three paths in the second reaction is C = S path < C = S path and C = O path < C = O path. In the reaction’s first step, the rate-determining step in both the single C = S path and the double C = S path possesses a higher barrier of 199.9 kJ/mol however, the 127.9 kJ/mol barrier in the double C = S path is lower than the 142.8 kJ/mol in the single C = S path. Hydrogen migration is crucial to the mechanism of CS 2 hydrolysis. Through optimizing the structure of reactants, intermediates, transition states, and products, it can be seen that the first step of CS 2 is that CS 2 reacts with H 2O first to form COS intermediate and the second step is COS intermediate reacts with H 2O to form H 2S and CO 2. Significant safety data come from the viscose rayon Industry, where both carbon disulfide as well as small amounts of H 2S may be present.Density functional theory (DFT) is used to look into the two-step hydrolysis mechanism of CS 2. In 2007 Alfa Aesar stopped selling carbon disulfide samples.Īt very high levels, carbon disulfide may be life-threatening because it affects the nervous system. Dilution with nitrogen rendered contents nonflammable. Johnson Matthey's sister company Alfa Aesar was the first company to introduce carbon disulfide in the form of pressurized bottle containing a solution of pressurized nitrogen, coupling agent, stablizer, and carbon disulfide, with an active carbon disulfide content of 85%. Worldwide exports of this chemical are negligible. Commercial AvailabilityĬS 2, being highly flammable and having one of the lowest autoignition temperatures, cannot be transported easily using commercial means. This conversion proceeds via the intermediacy of thiophosgene, CSCl 2.ĬS 2 is a ligand for many metal complexes, forming pi complexes. Sodium reduces CS 2 to give the heterocycle "dmit 2-": 3 CS 2 + 4 Na → Na 2C 3S 5 + Na 2Sĭirect electrochemical reduction affords the tetrathiooxalate anion: 2 CS 2 + 2e - → C 2S 4 2- ChlorinationĬhlorination of CS 2 is the principal route to carbon tetrachloride: CS 2 + 3 Cl 2 → CCl 4 + S 2Cl 2 Both xanthates and the related thioxanthates (derived from treatment of CS 2 with sodium thiolates) are used as flotation agents in mineral processing. This reaction is the basis of the manufacture of regenerated cellulose, the main ingredient of viscose, rayon and cellophane. Nucleophiles such as amines afford dithiocarbamates: It is widely used in the synthesis of organosulfur compounds such as Metham sodium, a soil fumigant. These differences in reactivity can be attributed to the weaker π donor-ability of the sulfido centers, which renders the carbon more electrophilic. Although it is isoelectronic to carbon dioxide, CS 2 is highly flammable:Ĭompared to CO 2, CS 2 is more reactive toward nucleophiles and more easily reduced. The reaction is analogous to the combustion of methane. A lower temperature reaction, requiring only 600 ☌ utilizes natural gas as the carbon source in the presence of silica gel or alumina catalysts: CH 4 + 1/2 S 8 → CS 2 + 2 H 2S CS 2 once was manufactured by combining carbon (or coke) and sulfur at high temperatures. Small amounts of carbon disulfide are released by volcanic eruptions and marshes. 3.1 Pressurized Liquid Nitrogen Based Sample. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
